Gilchrist Lab Projects
An organism in the natural world experiences daily and seasonal cycles of light, temperature, and humidity. Overlain on these regular patterns are random fluctuations and persistent changes in climate wrought by natural and human-driven alterations of habitat and atmosphere. In turn, these variations in climate dynamically alter the distribution of critical resources, change the demography of populations, and exert variable selection pressures over space and time. We seek to understand how this dynamic arena sets the stage for the evolution of ecologically important traits, addressing the following major questions:
- How do organisms adapt to a changing climate?
- What rates of climate-driven adaptive evolution can be obtained?
- How repeatable is that evolution?
- Does thermal adaptation play a significant role in biological invasions?
- What is the genetic basis of thermal adaptation?
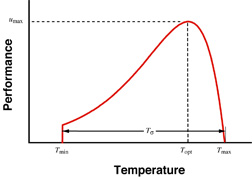
My theoretical and empirical work on the evolution of performance curves and reaction norms (Gilchrist 1995; Gilchrist 1996; Gilchrist 2000; Gilchrist and Kingsolver 2001) frames the research in my lab. A performance curve (illustrated at right) depicts the dynamic changes in fitness-related performance in response to a continuously varying external factor, such as temperature or photoperiod. Reaction norms usually refer to an irreversible change in morphology. Our understanding of the genetic basis and evolvability of phenotypic flexibility, either in terms of reaction norms or performance curves, is rudimentary.

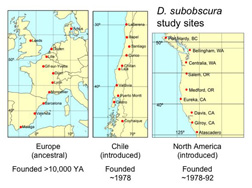
An Experiment in Nature: Evolution of ecologically important traits in invading and ancestral Drosophila subobscura.
The photo shows the pattern of wing size variation from Barcelona SP (left) to Aarhus DK (right). In the ancestral European populations, wing size increases with latitude. This species arrived in the New World in the late 1970's, setting up a classic experiment in evolution.
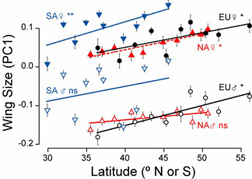
We found remarkable parallels in genetic markers, wing size, and some physiological traits that had evolved since the introduction in the New World. In the figure at the right, I show the first principle component (a multivariate measure of overall size: more positive values means bigger over several different body dimensions) as a function of latitude. These flies were reared in unform conditions for several generations ("common garden experiment"), so the clinal patterns represent genetic differences among populations. Females (the top three lines) all show parallel size clines, with larger body size in the north. Males (the bottom lines) slow clinal patterns in Europe and SA, however not in NA. Students in my lab are working to understand this change.
Finding the genes that matter: laboratory and natural evolution of high temperature thermotolerance in Drosophila melanogaster
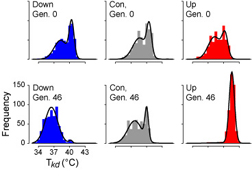
The rich array of genomic resources available for Drosophila melanogaster make it ideal for manipulative experiments in the lab that predict patterns that are testable in natural populations. In collaboration with molecular geneticist David Rand (Brown), we have examined flies that have been artificially selected for increased knockdown temperature , the temperature at which flies lose the ability to cling to a steeply inclined surface. Some typical KD temperature profiles are shown in the figure at upper left (after Gilchrist and Huey 1999), with the top row showing the profiles before selection and the bottom after 40+ generations of selection for decreased (in blue) or increased (in red) KD temperature. The "prostratometer" pictured at left is a Weber column modifed with a water jacket that allows us to precisely and quickly vary the internal temperature.
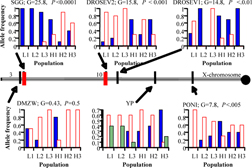
We have now identified “high temperature” QTL near bands 3 and 10 of the X chromosome (figure below) that are also present in other, independently derived lines selected for high temperature thermotolerance (Rand and Gilchrist in preparation). Our recently funded research uses fine-scale SNP and deletion mapping to identify candidate loci for further testing. As this work is just beginning, I have little more to report at present, however we plan several tests of our findings to date. One is to take the lab results back to the field, to examine clinal variation in the candidate loci along two latitudinal gradients in Australia and North America. We are currently constructing SNP maps for flies along the Aussie cline and will soon begin phenotyping them. A second test involves creation of new selection lines from Australian populations to further test the hypothesis that these X-linked genes are essential to the evolution of high temperature tolerance. We will travel to Australia within the next year to collect stocks and begin these studies.

We also use techniques, such as chill-coma tolerance (David et al. 2003), to examine physiological trade-offs in thermal sensitivity. Results obtained by Jennifer Hawley, a high school student working in my lab, suggest a positive correlation between high temperature tolerance and chill coma resistance in the knockdown selection lines. This surprising result suggests that the changes in these lines may be related to a general mechanism of stress tolerance. We have also begun screening mutants at some candidate loci near the third band of the X chromosome (period, timing, white) for signature differences in knockdown temperature. Ultimately, we will use the genes identified in this study to examine clinal variation in thermal performance of other Drosophila species, including D. subobscura.
We have just acquired a Sable Systems CO2/O2 analysis system and are starting to study the evolution of metabolic rate performance curves in both the lab selected lines and in D. melanogaster from clines in NA and Australia. Like D. subobscura, D. melanogaster is an invasive species, having left Africa some 300 years ago with human settlers to colonize its current range of natural populations. Understanding how selection imposed along latitudinal clines has shaped thermal sensitivity provides information crucial for predicting how populations will adapt to a changing climate. My postdoc on this project, Donna Folk, has a background in Drosophila environmental physiology. The collaboration blends my experience in field biology and laboratory selection, Rand’s skill as a molecular geneticist, and Folk’s knowledge of physiology to dissect the genetics and functional mechanisms that evolve as animals adapt to higher temperatures.
Other Projects
Additional projects our my lab involve cross-generational and inter-sexual effects on the evolution of thermotolerance. Previous work on parental and mating temperature effects (Huey et al. 1995; Crill et al. 1996) has led us to develop a new set of experiments examining trade-offs between sexually selected traits and stress tolerance. We are currently exploring the possibility of sexual conflict having a marked effect on the stress resistance of female D. melanogaster. We have observed a 30-40% reduction in mated females’ ability to survive a heat shock, relative to virgins. The effect is greatest when females are pre-treated with a sensitizing shock that induces the production of Hsp70, one of the major heat shock proteins. We hypothesize that the initiation of oogenesis may consume resources that unmated females use to make extra Hsp70, effectively limiting a mated female’s ability to mount a stress response. Certain male accessory gland secretions greatly accelerate oogenesis by diverting resources that females would use for maintenance into reproduction. Females have also evolved counter-measures that limit the action of the male accessory gland secretions; perhaps this defense itself is costly. One cool thing about D. melanogaster is that a large number of mutants are available to test various hypotheses about the cost of mating in terms of the stress response. A larger question is whether or not this association between stress tolerance and reproductive traits is a general feature of organisms.
Many of the data we collect depict complex reaction norms or performance curves, for which analytical techniques are still being developed (Gilchrist 1996; Gilchrist and Kingsolver 2001). I am involved with a NSF-funded collaboration with Richard Gomulkiewicz, Patrick Carter, and others to improve our understanding of these function-valued traits. Both empirical and statistical techniques are being developed to allow one to estimate genetic variances and covariances of functions (like the equations describing the curve in Figure 1) rather than working with the non-independent data points used to estimate the curve. I am collaborating with several members of this group on various projects.
A culture and community for research
I think of my lab and those doing related research as a community. All of my grants have travel money for meetings of collaborators because it is essential to get all of our heads together after the data collection is over. We meet with the Huey lab once or twice a year. My postdoc, Donna Folk, is expected to spend 3/4 of her time in my lab and 1/4 at David Rand’s at Brown. We plan semi-annual gatherings for the PI’s, students and techs involved in the project to share techniques, data, and insights. The function-valued trait grant sponsors semiannual meetings for the participants. Locally, my lab sponsors a variety of events, such as “Paella Night”, “The Fall Zinfandel Fiesta”, “Do-It-Yourself Sushi”, etc. to build community. Successful collaborations require both technical and social skills; I train students in these skills because much of the significant biological research today and in the future will require teamwork and openness to interdisciplinary approaches. In other words, we work hard, but we have fun, too…